Vector control and Isolation of Larvicidal Compounds from Melia azedarach (A. Juss) Leaf using Anopheles gambiae Larva.
Keywords:
Melia azedarach, β-sitosterol, Triterpenoids, Larvicidal activityAbstract
Background: Despite ongoing efforts to control the disease, malaria remains a serious threat to the human race on earth. This disease is ubiquitous in Africa, especially those countries that are categorized as undeveloped. The present investigation aims to evaluate and isolate larvicidal principles in the leaves and stem bark of Melia azedarach from the Meliaceae family using the fourth instar larvae of An. gambiae.
Methods: Larvicidal activity was evaluated by exposing 4th instar larvae of Anopheles gambiae to serial concentrations (0.625-10 mg/mL) of extracts, fractions, and compounds of Melia azedarach. Larval mortality was recorded after 24h of exposure and 50% lethal concentration (LC50) values were determined. LResults were compared to those of larvae exposed to N, N- diethyl-m-toluamid (DEET), the reference insecticides, and untreated groups.
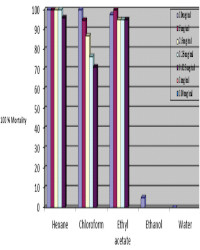
Results:Larvicidal activity of the crude methanol extract of both leaves and stem bark of M. azedarach were 5.80 and 7.59 mg/mL respectively. The hexane soluble fraction of M. azedarach leaf displayed highly lethality at 0.625mg/mL with LC50 of 0.025 then the remaining fractions. Similarly, larvicidal activity of DEET was signifificantly low (LC50 = 1.09 mg/mL) than the hexane fractions (p < 0.05). Fractionation and purifification of the active fractions led to the isolation of three compounds. Sub fraction 35-49 (designated as MAZLFF) of M. azedarach showed larvicidal activity on larvae with LC50 of 0.19 mg/mL. Two compounds were isolated from MAZLFF using preparative thin layer chromatographic. Compounds 2 and 3 had Rf values of 0.75 and 0.63 respectively on silica gel G using CH3Cl3 :CH3OH (9.5: 0.5) and CH3Cl3 : Acetone (9:1) re spectively. Compounds 2 and 3 showed a positive test for the presence of triterpenoids after spraying with anisaldehyde/H2SO4 . The presence of triterpenoids may be responsible for the insecticidal activity observed in the two compounds isolated from the leaves of M. azedarach.
Conclusion: The leaves of M. azedarach showed potent anti larvicidal activity. Compounds from this plant may serve as sources for the development of malaria vector control compounds.
References
World Health Organisation (WHO). (2018). World Malaria Day 2018; Ready to Beat Malaria.
Ranson H, Rossiter L, Ortelli F (2001). Identification of a novel class of Insect Glutathione S-transferases involved in resistance to DDT in the Malaria Vector Anopheles gambiae. Biochemical Journal 359: 295 – 304. https://doi.org/10.1042/0264-6021:3590295
Curtis CF (1994) Should DDT continues to be recommended for malaria vector control? Medical Entomology 8: 107 – 12
https://doi.org/10.1111/j.1365-2915.1994.tb00147.x
Collins FH (1995). Paskewitz SM. Malaria: current and prospects for control. Annual Review of Entomology. 40: 195 – 219. https://doi.org/10.1146/annurev.en.40.010195.001211
Feinstein L (1952). Insecticides from plants. In: Insects: The year book of agriculture, U.S.D.A. Washington, D. C. 22-229.
D'Ambrosio M and Guerriero A(2002). Degraded limonoids from Melia azedarach and biogenetic implications. Phytochemistry 60: 419– 24. https://doi.org/10.1016/s0031-9422(02)00107- 3
Nakatani M, Abdelgaleil SAM, Saad MMG (2004). Phragmalinlimonoids from Chukrasia tabularis. Phytochemistry 65: 2833–41
Banchio E, Valladares G, Defago M (2003). Effects of Melia azedarach (Meliaceae) fruit extract on the leaf miner Liriomyza huidobrensis (Diptera: Agromyzidae): assessment in laboratory and field experiments. Journal of Annual Applied Biology 143: 187– 93.
Wandscheer CB, Duque JE, da Silva MAN(2004). Larvicidal action of ethanolic extracts from fruit endocarps of Melia azedarach and Azadirachta indica against the dengue mosquito Aedes aegypti. Journal of Toxicology 44: 829–35. https://doi.org/10.1016/j.toxicon.2004.07.009
Bezanger-Beauquesna. (1938). The Useful Plants of West Africa. Second Edition, 141, 54 (1938). https://doi.org/10.1038/141054a0
Kingsbury JM. (1964). Poisonous Plants of the United States and Canada. Prentice-Hall, Englewood Cliffs, NJ 206-208.
Macial MV, Morais SM, Bevilaqua CML (2006). Ovicidal and Larvicidal activity of Melia azedarach extract on Haemonchons contortus. Journal of Veterinary Parasitology 140:98-104. https://doi.org/10.1016/j.vetpar.2006.03.007
World Health Organization (WHO), (1997). World malaria situation in 1994. Wkly Epidemiol Rec67: 161 - 167. https://doi.org/10.1590/S0102-695X2009000100026
Ranson H, Rossiter L, Ortelli F, Jensen B, Wang X, Roth C.W, Collins FH, Hemingway J. (2001). Identification of a Novel class of Insect Glutathione S-transferases involved in Resistance to DDT in the Malaria Vector Anopheles gambiae. Biochemical Journal. 359: 295 – 304. https://doi.org/10.1042/0264-6021:3590295
Elimam, AM, Elmalik KH, Ali FS. (2009). Efficacy of leaves extract of Calotropis procera Ait. (Asclepiadaceae) in controlling Anopheles arabiensis and Culex quinquefasciatus mosquitoes. Saudi Journal of Biological. Sciences., 16: 95 - 100. https://doi.org/10.1016/j.sjbs.2009.10.007
Koul O, Jain MP, Sharma VK. (2000). Growth inhibitory and antifeedant activity of extracts from Meliadubia to Spodopteralitura and Helicoverpaarmigera larvae. Indian Journal of Experimental Biology 38(1):63-8.
Coria C, Almiron W, Valladers G (2008). Larvicidal and oviposition deterrent effects of fruit and leaf extract from Melia azedarach L. on Adese aegypti (Dipetra: Culicidae). Bioresources Technology 99 (80): 3066 - 70. https://doi.org/10.1016/j.biortech.2007.06.012
Govindachari TR, Krishna Kumari GN, Gopalakrishnan G (2001). Insect antifeedant and growth-regulating activities of quassinoids from Samadera indica. Fitoterapia; 72: 568-571. https://doi.org/10.1016/s0367-326x(00)00342- 7
Bohnenstengl FI, Wray V, Witte L (1999). Insecticidal meliacarpins (C. Seco limonoids) from Melia azedarach, Phytochemistry 50: 977 - 982. https://doi.org/10.1016/S0031-9422(98)00644-X
Marise MOC, Esteban RFC, Paloma MM, Celma MSG, Vanderleia CO, Alphonse K (2008). Melia azedarach extracts and their activity on Musca domestica L.Revista Brasileira de Fermacognosia 18 (suppl).
Usta J, Kreydiyyeh S, Bakajian K. (2002). In vitro effect of eugenol and cinnamaldehyde on membrane potential and respiratory chain complexes in isolated rat liver mitochondria. Food Chemistry Toxicology 40: 935 - 940. https://doi.org/10.1016/s0278-6915(02)00071- 6
David JP, Rey D, Pautou MP (2000). Differential toxicity of leaf litter to dipteran larvae of mosquito development sites. Journal of Invertebrate Pathology 75: 9 - 18. https://doi.org/10.1006/jipa.1999.4886
Published
How to Cite
Issue
Section
License

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.


